As the winter chill settles in over the northern states, the boat winterizing process is in full swing. If you are about to winterize your boat for storage, or have already winterized and worry about having the right antifreeze solution, there are a couple surefire ways to find out. Having the right antifreeze solution matters for two reasons: first, you don’t want your boat’s plumbing or engine cooling system to freeze or corrode; second, you don’t want your potable water system to turn into a biological breeding ground this coming spring.
A few years ago, while the polar vortex was pummeling the northern states, Practical Sailor tech editor Drew Frye was knee deep in glycol antifreeze and engine coolants. The range of tests he ran compared how well the myriad products on the market protect our water supply and engine-cooling systems from freezing, corrosion, and biological growth. He reported the results in the September 2014 issue of Practical Sailor, which featured in-depth look at antifreeze.
Although we found some clear differences between the various antifreeze products on the market, one of our most important findings was that how you use antifreeze is as important as what product you use. Even the best product, mixed with too much water left in the line, results in a blend with unknown and perhaps unsatisfactory performance. While this may not be critical in North Carolina, sailors in Wisconsin need to get it right.

There is only one way to do that; measure the glycol as it comes out the other end of the plumbing. A $10 hydrometer from the local auto-parts store will do this. You have to collect a suitable volume of fluid in a cup, and if there is any oil (which floats) or dirt (which sinks), the reading will be affected. Remember to first check the calibration with water; if its off, buy a new one. In the end, you can guess based on color.
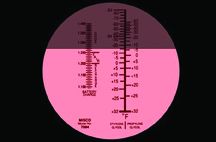
Or you can buy a tool that will last a lifetime, can be calibrated (small hidden screw on the bottom side) to read within 1 degree of the true freeze point, using only a few drops, and will even read the state-of-charge on your battery’s wet cells. We are talking about the pocket refractometer, which looks like a small spotting scope.
How does it work? In addition to changing the density of water, glycols, alcohols, and acid affect the way light is bent, and a refractometer can measure this change. To use it, the user places a few drops of the target liquid on the prism, closes the cover, looks in the eye piece, and sees a sharp line that can be read in seconds.
Pocket refractometers sell for $75 to $150 through many online auto-parts retailers.
For more information on this topic, I have addressed coolants and antifreezes in two previous blog posts. In one report, I cautioned against using propylene glycol in engine-cooling systems that have acetal plastic (often used in clear sight bowls for raw-water intake filters). In the other, we looked at the relationship between glycol content in your antifreeze and the level of protection against harm from freezing.











Hmmm – for water system simplify with RV antifreeze (pink) which is non-toxic. And I have always heard that 50-50 is as good as it gets and you can buy that pre-mix.