Photos by Drew Frye

Protecting marine water systems from freeze damage is a deceptively simple goal. The terminology and various product claims can be confusing, and what seems like a good common-sense decision can lead to trouble.
We tend to think that all water systems are the same; that boats as well as RVs can be protected by the same pink antifreeze without any further thought. However, many of the problems we associate with age, or normal wear and tear-stiff impellers, cracked pipes, ruined joker valves, and foul-tasting tap water-can often be attributed to errors during winterization.
Winterizing products for potable water systems are most commonly formulated using one of two chemicals: propylene glycol (PG), a relatively non-toxic liquid similar to glycerin, but less viscous; or denatured ethanol, which is generally less expensive and performs much the same function, preventing freezing and bursting. In denatured ethanol, a small amount of a denaturant (a foul tasting and often slightly poisonous chemical) is added to prevent people from drinking it. Ethylene glycol (EG) is common in engine coolants but is not appropriate for potable water systems because of significant acute toxicity. Ethylene glycol is appropriate only for engine and black-water systems, where there is no risk of accidentally ingesting it.
What we tested
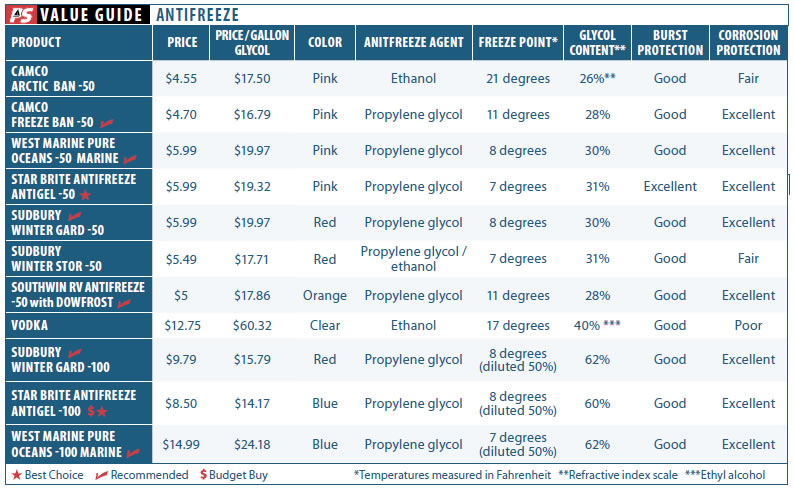
We tested a range of products, many of them marketed to both the RV and marine markets. While many manufacturers sell the same product to each of these markets under a different label, antifreeze seems to be one of those products which are jointly marketed to both crowds. The test field included products from Star brite, Sudbury, Camco, and West Marine.
Freeze point vs. Burst Point
To decipher antifreeze ratings, it helps to understand the difference between freeze point and burst point, the latter being the temperature protection that is most often promoted on a bottle of antifreeze.
Burst point is a fuzzy term. There is no ASTM or other industry-accepted standard procedure to determine it. In principle, burst point is the temperature at which enough of the solution is frozen that instead of contracting with continued cooling, it begins to expand. But how much expansion and how much force constitutes a burst point?
The standard used for antifreeze is based upon copper pipe, but boat systems include pipes, hoses, filters, and reservoirs made of plastics that become brittle and much weaker than copper in cold temperatures. Two antifreeze manufacturers-West Marine and Star brite-communicate burst point differently on their labels. They offer guidance based on the warmer burst points for PVC pipe, typically -10 degrees instead of the -50 degrees quoted for copper pipe.
Curiously, water is one of very few materials that expands when freezing. (Thats why ice floats.) By contrast, most other materials, including glycols, contract and sink when they freeze. Once the solution reaches a certain glycol content, bursting becomes nearly impossible because the solution cannot expand upon continued cooling.
Generally, products that claim protection up to -50 degrees contain about 30 percent glycol. Products with -100 protection generally contain 60 percent glycol, and products with -200-degree protection generally contain more than 90 percent glycol. Before making any assumption regarding a products level of protection, you should check the makers specs or measure the freeze point of dilutions.
Observations
We carried out a number of tests to evaluate everything from burst point to corrosion protection and compatibility with pump components. (See How We Tested.) The testing included subjecting product-filled vials to below-freezing temperatures, and submerging metal coupons in the products to evaluate corrosion protection.
After two months of immersion, most of the corrosion-test samples looked the same as when testing began; others looked remarkably different. The aluminum coupon submerged in tap water was very corroded. The vodka sample de-zinced brass, tarnished copper, and pitted aluminum. The low-end products from Camco (Arctic Ban) and Sudbury (Winter Stor) did not protect aluminum. The balance of the products were impressive in their protection against corrosion, leaving pristine coupons. Look for a more in-depth report on corrosion protection in a follow-up article on engine coolants.
In our past tests on joker valves, we found that some valve materials were more vulnerable than others to antifreeze (see PS July 2013 online). Propylene glycol is rough on neoprene (Jabsco joker valves); ethanol is rough on nitrile (most other brands); and both are questionable with PVC (both rigid pipe and flexible hose), polycarbonate, and acetal. Ethylene glycol is easier on all of these materials, but it cannot be used in potable water systems.
Although none of the products in this test field is suitable for use as an engine coolant, we did place several of them (Southwin RV Antifreeze, Star brite -50, Pure Oceans -50) in engine coolant corrosion test rigs (ASTM D1384, corrosion in glassware). We found them to be many times better than tap water in protecting against corrosion and nearly as effective as engine coolants. These should be quite safe for use in winterizing sea-cooled engines.
The Proper Mix
Repeated freeze-thaw cycles tend to separate glycol from water (water-ice floats), effectively weakening your antifreeze concentration. In our view, it is cheap insurance to protect expensive systems like engines to the freeze point rather than the burst point. This requires only 10- to 20-percent higher glycol levels.
In a 0-degree climate, let an undiluted -50-degree burst-point concentration run through your system until about +10 degrees is obtained, and in a -30-degree climate, consider using the -100-degree glycol, running it through to get a freeze point around 0 degrees.
What about the toxicity of ethylene glycol toward the environment? A sorry bit of urban legend perpetrated by the makers of ethylene and propylene glycol products. We posted a long explanation, backed by both the U.S. Environmental Protection Agency (EPA) and international studies, showing that neither of these formulas have significant toxicity toward marine organisms and that they are equivalent (see Inside Practical Sailor, June 10, 2013). Both can be recycled, though do try to keep as much water out of the waste as possible; recyclers tell us that they prefer at least 25-percent glycol.
Camco Arctic Ban -50
A slightly lower priced product based on denatured ethanol, the Camco Arctic Ban -50 performed well enough in burst-point testing, but not so well in corrosion protection tests. We don’t see enough difference in price justify its use over the better performers in this test.
Bottom line: Testers deem it a less-expensive product for a low end market.
Camco Freeze Ban -50
Camcos propylene glycol-based Freeze Ban -50 RV and Marine Antifreeze -was one of the better performers in our burst-point testing. Corrosion protection was excellent.
Bottom line: Testers Recommend the Freeze Ban as a budget-friendly option for -50-degree protection.
Southwin RV Antifreeze -50
The Southwin RV Antifreeze -50 is a propylene glycol-based product that also contains Dowfrost additive. Dowfrost is perhaps the best-known corrosion inhibiting PG-based heat transfer fluid in the U.S.. It is ubiquitous in the commercial refrigeration and air-conditioning industries, so it wasnt a surprise that the Southwin RV Antifreeze was rated Excellent in corrosion-protection tests. It also performed very well in burst-point testing.
Bottom line: Southwin RV Antifreeze -50 earned our Recommendation.
Star brite -50 Antifreeze Antigel
A premium propylene-glycol product, Star brites -50-degree Antifreeze and Antigel, designed for marine and RV use, provided the best freeze and burst-point protection of the group. It was typically the last to freeze and showed the greatest glycol content, if only by a tiny measure. It also performed very well in corrosion testing. The pink liquid is formulated to be used without being diluted.
Bottom line: This is the Best Choice for burst-point protection to -50 degrees.
Star Brite -100 Antifreeze Antigel
A more concentrated version of the Star brite -50, the -100-degree Antifreeze and Antigel has twice the glycol-perfect for boats in extreme climates. The extra glycol allows for inevitable dilution with undrained water. For the price conscious, it also offers an opportunity to save a few dollars; simply split the contents into two matching jugs, halving it; then fill with water, and you have two gallons of -50 burst-point winterizing fluid.
Bottom line: For those seeking -50-degree protection, its the Budget Buy, so long as youre willing to do a little extra mixing. Among the -100-degree products, its the Best Choice.
Sudbury Winter Gard -50
A top-shelf propylene-glycol product, Sudburys Winter Gard -50 antifreeze performed very well in burst-point tests and Excellent in anti-corrosion testing.
Bottom line: Another tester favorite, its Recommended.
Sudbury Winter Gard -100
Like the Pure Oceans and Star brite -100 products, Sudburys Winter Gard -100 contains twice the glycol content of the -50 Sudbury Winter Gard. This makes it suitable for colder climates and economical in warmer climates if diluted 50/50 with water. The propylene-glycol product performed very well in burst-point and anti-corrosion testing.
Bottom line: Testers Recommend it.
Sudbury Winter StoR
The Sudbury Winter Stor RV and Marine Antifreeze is an ethanol/propylene-glycol blend that provided good burst-point protection in tests, but was disappointing in the area of corrosion protection. (This is why we don’t prefer ethanol-based products.)
Bottom line: The premium products are worth the added cost, compared to this price-point product.
West Marine Pure Oceans -50
West Marines Pure Oceans -50 Marine Antifreeze is a traditional propylene-glycol product that performed well in testing.
Bottom line: Testers Recommend it.
West Marine Pure Oceans -100
A more concentrated version of the Pure Oceans -50 antifreeze, the -100-degree formula has twice the glycol. It is perfect for boats in extreme climates. As with the other -100-degree blends, the extra glycol allows for inevitable dilution with undrained water, or n more moderate climates, you can mix it 50/50 with water.
Bottom line: We Recommend it. Its a good value, if you don’t mind mixing.
Skol Vodka
A bottom-shelf spirit, cheap vodka is often touted as an alternative to conventional winterizing agents. The primary positive that some tout-which we determined to be untrue during testing for a future article-is that it leaves less aftertaste in the plumbing than glycols. Weak, fermented vodka made the most disgusting mix.
Where vodka falls down is in the price category. In the U.S., it is more than four times more expensive than the least expensive propylene-glycol alternative. We also worry about making sure we get the right dilution when mixing with water in the system.
Bottom line: Not Recommended.
Conclusions
All of the test products were effective freeze-proofing agents if used properly, with some small variation in glycol/alcohol concentration. Which is the best value? The Star brite and West Marine Pure Oceans products, because they have slightly higher glycol concentrations and tend to offer the better value. In general, -100-degree products offer more glycol for the dollar, if you don’t mind pouring one gallon of product into two jugs and shaking. The more concentrated products (-100 burst point) also give you the option of recovering that weak first flush, mixing it with something strong, and adding it to other systems, further reducing both cost and waste. Be sure to always check the freeze-point/burst-point chart for your product and use a worst-case winter when projecting the temperature ranges your boat will have to endure.
We recommend against winterizing with solutions containing alcohols. Corrosion protection of all ethanol-based products was poor. Field measurement of freeze point requires a refractometer and a conversion chart or ethanol hydrometer. To make matters worse, these results are not accurate if the product has been mixed with glycol. We worry about alcohol and ethanol evaporation, particularly if the vessel is serviced early in the fall. The bottom line is that the small price advantage does not justify their use.
For head systems and engines, consider ethylene-glycol antifreeze. It offers better materials compatibility for neoprene and nylon. For potable water systems, use propylene glycol.
The key to success, however, is proper usage. Drain the water tank and water heater; add the antifreeze only to the plumbing (not the tank). Get a refractometer and test the glycol draining from the far end of the system-be certain you actually have the same mixture you think you do. We found color to be a poor indicator. Protect the system to near the freeze point, not the burst point.
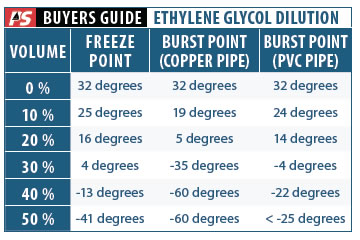
This table shows the burst point and freeze point for various concentrations of ethylene glycol; it is based on manufacturer data, supplemented with some of our own experiments. Colder temperatures require higher concentrations of ethylene glycol, up to 50 percent. For maximum protection, PS recommends using freeze-point temperatures to calculate the antifreeze dilution.