250
Practical Sailor has twice looked at fuel additives in recent years. In November 2008, we looked at the ability of fuel additives to prevent the potentially damaging water “drop out,” or separation, that occurs in E-10 fuels (gasoline that has 10-percent ethanol content) that are stored over long periods. In July 2009, we looked at diesel fuel additives that claim to combat biological growth. In this test, we again look at gasoline additives, focusing on the ability to prevent corrosion, which can lead to damaging metal and solid deposits in the fuel system.
Anyone who has had to rebuild the carburetor on a small outboard is familiar with their affinity for choking on the smallest of deposits or fine solids. In diesel engines, these deposits appear in the fuel tank over time, the result of fuel oxidation, dirty fuel, and bacterial growth. But significant buildup in gasoline tanks is unusual. In the case of diesel engines, multi-stage filtration is generally sufficient to catch the solids. In gasoline engines, however, solids often appear spontaneously in the carburetor bowl, no matter how many filters are employed.
For this test, PS looked at composite samples taken from four engines, none of which had used aftermarket additives: two 2001 Yamaha 9.9-horsepower four-stroke engines, one 2006 Mercury 6-horsepower four-stroke, and one 2002 Mercury 3.5-horsepower two-stroke. We found the following contaminants, expressed as a percentage of total weight: aluminum, 21 percent; zinc, 8 percent; calcium, 10 percent; iron, 5 percent; copper, less than 100 parts per million (ppm).
The mere presence of a metallic element in the solids does not prove that they came from corrosion. These solids could have been introduced during refining or distribution and then deposited in the fuel tank over the course of years of evaporation cycles, but given the typical ash content of fuel, this seems highly unlikely. There are also polymer additives that can dry out, forming a gel. Some studies have reported sodium salts in fuel.
The presence of aluminum warrants special discussion. Both fuel tanks and carburetors often contain aluminum, and the gasoline additive ethanol has been linked to aluminum corrosion. Without delving too far into the chemistry, ethanol behaves as a weak acid, attacking the oxide coating that normally protects aluminum from corrosion. This exposes a reactive aluminum surface that the dissolved oxygen found in water can quickly oxidize. In the absence of oxygen, ethanol itself can serve as the oxidant, though this process is much slower.
If conditions are right, this two-step chemical process produces pitting that can, over time, bore holes through a fuel tank. Corrosion byproducts can clog small passages. Corrosion inhibitors, often added during refining, will slow this process to a crawl. We wanted to find out whether after-market additives added further protection.
There is also galvanic corrosion to consider. All of the carburetors tested contained some brass parts, and the Yamaha engines have brass drain plugs. The difference in potential between aluminum and brass on the galvanic scale is 0.45-0.5 volts, depending on the specific alloy; brass and aluminum are well known to be reactive when placed in contact. In dry fuel, however, there is no conductivity and no corrosion, so before the introduction of E-10 fuel, galvanic corrosion was a small issue. E-10, however, can dissolve significant atmospheric moisture and saltwater in small amounts, and this water is transported throughout the fuel system. In this way, E-10 delivers the key ingredient for fuel system corrosion: water.
Testing Corrosion Resistance
Laboratory corrosion testing is a very well-developed science. The American Society for Testing and Materials (ASTM) and National Association of Corrosion Engineers (NACE) have volumes on these methods. However, their focus is different than what we would like to achieve. Laboratory methods generally focus on product development and quality control. Test conditions are made severe, so the results can be obtained within a few weeks at most. This allows labs to quickly and economically sort through candidate formulations and test product batches. These accelerated methods can’t always accurately model the slow processes and chemical breakdowns that occur over months and years. They often force failures that don’t actually happen and overlook more subtle but realistic failures modes.
For example, in 1995, General Motors introduced a revolutionary product, Dexcool. While it had been the subject of extensive laboratory and fleet evaluation, they never tested it with certain gasket materials nor mixed with any other coolant. As a result, many cars suffered gasket failures and any topping off with other engine coolant was a disaster. The coolant has since been reformulated.

450
How We Tested
Our original goal was to run a long-term test of fuel additives. Instead of adding heat or oxygen or excessive corrosive materials, we would substitute time. In the interests of gathering controlled repeatable data, we took a laboratory-style approach that incorporated methods used for testing engine coolants and fuel additives. The nuts and bolts of the testing procedure are on page 17.
Initially, we tested E-10 gasoline without any water or additive additions, but after six months of exposure in humid conditions, we found no measurable corrosion. The corrosion inhibitors in the retail E-10 were effective in preventing damage. If you are meticulous about your fuel (see “Maintenance Tips,” page 19) and store it for no more than six months, there should be no need for an additive. However, we know corrosion occurs.
Thus, we elected to add a small amount of seawater—only 0.03 percent (the equivalent of 3/4 of an ounce of seawater in a 20-gallon tank, an amount E-10 can easily hold in solution)—to hurry things along. It is our belief that serious corrosion in carburetors is rare and probably only occurs when either a lot of atmospheric water or trace seawater mist sneaks into the tank. That’s when a carburetor needs protecting.
Our decision to accelerate testing focused on the ability of additives to resist salt-induced galvanic corrosion, a scenario that some additive makers criticized as unrealistic, one that the products were not designed to combat. We’ve included maker comments regarding the test under the related section for each product.
It is possible that our final rankings would be different if we considered only the corrosion agents in the E-10. It is also fair to say that those additives that experienced only minor failures are likely to be adequate under normal conditions. However, since all samples were exposed to the same harsh conditions, we believe our relative rankings are good guide for consumers—particularly when one considers the risks of contaminated fuel for a cruising sailor.
It is our hope that as the marine fuel additive industry attempts to enhance standards for these additives, they will take a closer look at our test for new benchmarks.
We selected a bundle of standard, 1-by-2-inch, alloy coupons as used in ASTM method D1384—a glassware method for evaluating engine coolants that was modified to serve our test purposes. This approach ensured uniform metal alloys of known composition. Additionally, these coupon bundles are designed to mimic the galvanic corrosion effects common to machines assembled from dissimilar metals. The couplings used were aluminum/brass/steel (to mimic a carburetor bowl) and copper/steel/brass (to mimic a steel tank and fuel lines).
As stated, we preferred more realistic exposure to the atmosphere and a longer time period for testing. Standard methods require starting with gasoline and pure ethanol without any refiner additives, but we didn’t think that test, while fine for refiners, was valid for testing aftermarket additives used by boaters. We chose to test retail formulated E-10 (10-percent ethanol) 87 octane gasoline from a busy Shell Oil dealer.
What We Tested
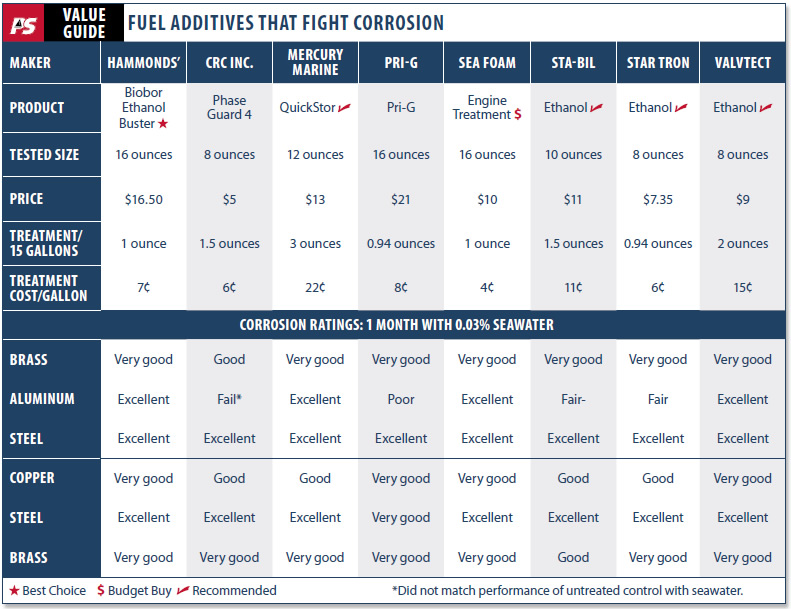
All of the products tested make essentially the same recommendations and claims: that the product should be used on a regular basis and specifically before long-term storage and that it prevents ethanol breakdown, gum formation, keeps carburetors and injectors clean, and promotes easier starting. All but one claim to prevent corrosion; the other implies this. We only tested the anti-corrosion assertion, and we only tested products marketed as storage additives. We dosed each additive according to label recommendations.
In all, we tested eight products from eight manufacturers, representing a cross-section of what is on the market: Hammonds Biobor Ethanol Buster; CRC Phase Guard 4; PRI-G; Mercury Marine Quickstor; Sea Foam; Sta-Bil Ethanol; Star Tron Ethanol; and ValvTect Ethanol.
Observations
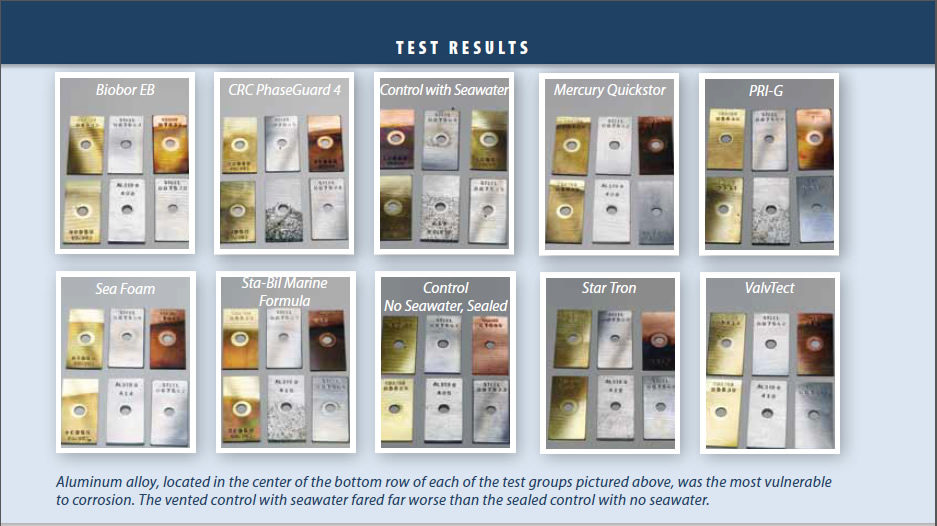
The sealed control sample appeared not to age at all, and there was no corrosion or staining of the metal coupons. Clearly, anything you can do to minimize water gain and breathing will help prevent corrosion.
The vented control sample without seawater addition showed virtually no corrosion after six months of exposure. Since the atmospheric water absorbed through the vent is distilled water, which has very low conductivity and does not encourage galvanic corrosion, the damage was minor.
The vented control with seawater addition showed fast corrosion, particularly on the aluminum coupon. Brass and copper showed heavy staining, and there were tiny pits on the steel coupons. The discoloration and pitting resembled that of the worst carburetors and fuel components we have seen in 15-plus years of marine service. Some mechanics we spoke with reported pin-holes in carburetor bowls, and our model seems to represent a realistic worst-case scenario.
Saltwater intrusion is clearly dangerous, even in minute amounts. Every effort should be made to insure that the filler cap is tight. There have also been reports of sodium salts in E-10 that could be similarly harmful, and as one maker suggested, have negative consequences when combined with an additive.
Inhibitors varied in their effectiveness. Even with salt additions, some were able to suppress nearly all corrosion. Some had no effect at all. We were surprised by the severity of the damage, and ran duplicates to confirm our findings; the duplicates showed no significant deviation from the originals.
Biobor EB
Biobor EB is made by Hammonds, the manufacturer of Biobor JF, which is the most widely used biocide for diesel and jet fuel, and a Recommended product in our diesel biocide tests. It seems clear the company has another winner here: Biobor EB (ethanol buster) is a top-performing additive with no metal damage and only the slightest staining in our tests. Biobor EB is available in 16- and 32-ounce bottles.
The maker commended our test report as “thorough and professionally conducted.”
Bottom line: Reasonably economical and clearly the top performer, this is our Best Choice.
CRC Phase Guard 4
CRC Industries claims its Phase Guard 4 helps suspend water and delay phase separation. Perhaps this is its downfall, as it performed poorly, with heavy aluminum pitting and heavy staining on copper and brass. It is available in 8-, 16-, and 32-ounce bottles.
Adam Seilisker, vice president of technology for CRC, took issue with our test in regards to the arbitrary choice of .03 percent saltwater solution and the small sample size, among other things. He said a test using dissimilar metals “introduces complex reactions that might not be reflective of the environment being studied. . . . We are confident that CRC’s Phase Guard 4 product meets all of the performance criteria for its intended purpose.”(Here is the full text of CRC’s statement: CRC Comments)
Bottom line: Not recommended.
Mercury Marine QuickStor
The Mercury Marine Quickstor turned out to be a very solid performer and is made by a respected outboard manufacturer. Quickstor showed a bit more copper staining than Biobor, but there was no metal damage, and it had very clean aluminum coupons. (Aluminum was consistently the most difficult metal to protect.) It comes in a 12-ounce bottle.
Bottom line: Quickstor was the most expensive of the group, and while we recommend it for its solid performance, there are less expensive alternatives.
PRI-G
Testers noted heavy staining and significant aluminum pitting in the PRI-G sample test. The product was shown to suspend water in prior Practical Sailor testing and seems to be damaging in the presence of seawater. It is available in 16-ounce, 32-ounce, and 1-gallon bottles.
Ralph E. Lewis, a vice president with PRI-G, questioned the level of contamination in our test, calling it “extremely rare,” but adding that “should a seawater intrusion occur, the level of contamination would be far higher than the amount you used.”
He pointed to the product’s extensive standards-based testing and record with commercial clients, including “more than 1,500 oceangoing vessels—both distillate and heavy fuels. . . . Practical Sailor might as well have designed a test to determine if PRI-G makes a good topping for vanilla ice cream.”
Bottom line: Not recommended.
Sea Foam
Sea Foam was a pleasant surprise, as it is not known as a marine storage additive, but rather as a treatment for hard-to-start engines. In fact, don’t look for it at the marine center at all, but rather at the neighborhood autoparts chain store. The Sea Foam test coupons were all very clean, and the fuel had very little color change from aging.
Bottom line: The most economical choice and a top performer. It is our Budget Buy.
Sta-Bil Ethanol
From Gold Eagle Products, Sta-Bil is perhaps the best known brand of all winter storage additives, and the special marine formula for ethanol fuels that we tested is widely available. This was our top-recommended additive for ethanol fuels in the 2009 test. While this product has a strong reputation as a gum-preventing storage additive, it did not perform as well as others in corrosion prevention in this specific test. It did help, though, and we believe it would perform well in the absence of seawater contamination. It is available in 8-, 16-, and 32-ounce dispenser bottles.
Bottom line: While it isn’t a leader for corrosion protection, it remains our top-rated ethanol treatment and is provisionally recommended for corrosion protection.
Star Tron Ethanol
Star Tron is made by Star brite, a company that markets a range of boat maintenance products, several of which have earned top ratings in Practical Sailor tests. We previously tested Star Tron for its ability to delay or prevent phase-separation in ethanol fuels, and its ability to burn cleanly without leaving behind any ash residue. In that test, the product came out with a Recommended rating.
This time around, testers found that Star Tron helped inhibit corrosion, but it didn’t stand out as a top performer. In fact, Star Tron doesn’t claim to use corrosion inhibitors, but rather focuses on keeping the system clean.
It helped, though less than others, and we believe it would perform well in the absence of seawater contamination. Star Tron is available in 8-ounce, 16-ounce, 32-ounce, and 1-gallon bottles.
An emailed statement from one of Star brite’s engineers (who Star brite prefers to keep anonymous) pointed out the value of a “do no harm” product in the tricky environs of marine fuels: “When succinic acid, carboxylate acids, amine salts, and ester-based anti-corrosive additives (that’s pretty much every additive on the market) mix with sodium, they form very sticky deposits. This kills High Pressure Common Rail diesels by sticking up the injectors. . . . Singularly stopping corrosion in seawater-contaminated gas is important, but treating the vast majority of fuel with unnecessary extra anti-corrosive additives (recall the refinery blend works fine for all but seawater-contaminated fuel) is not always the best answer.”
Bottom line: Based on this test and our 2009 test of ethanol additives, this product is Recommended, although its corrosion prevention is not as effective as others in the test.
ValvTect Ethanol Gasoline Treatment
The ValvTect product delivered very good protection and compared favorably with the Best Choice Biobor. The only reason this was not named a “Best Choice” was its higher cost. If you use this product for other reasons, there is certainly no reason to switch. It is available in 8- and 12-ounce bottles.
Bottom line: ValvTect Ethanol was a top performer. It is Recommended.
The Future of Fuel Additives
A group of manufacturers and additive producers has formed a working group in cooperation with the National Marine Manufacturers Association (NMMA) with the intention of setting standards for additives, identifying approved products, and sorting out those they find damaging. The working group has already determined that any additive that can suspend or dissolve significant amounts of water will be rejected. Engine manufacturers we spoke with recommended against using any alcohol-based products such as MDR E-Zorb that dissolve water so that it could be “cleanly burned” through the engine.
Conclusion
When it comes to corrosion prevention that will ensure a long engine life, drier and less oxygen are best. We reiterate our advice to avoid products that suspend water, which can lead to more corrosion-related carburetor clogging troubles as they help distribute water and salt throughout the fuel system.
Another element in the fuel protection chain is gas-tank filters, soon to be phased into new boats with built-in fuel tanks. We are currently testing a variety of filters, and preliminary results suggest that they may also be a helpful in protecting your fuel system, but it is still too early for us to know for certain.
We believe that E-10 corrosion issues should become less important as engine manufacturers move away from alloys known to be vulnerable to E-10 and as fuel-dispensing equipment at service station and fuel docks is updated. Specifically, zinc and Zamak die-cast parts must be avoided. Fuel producers are also getting smarter about which corrosion inhibitors to use. We believe we are already seeing some improvement.
Some additives really help, stopping corrosion dead in its tracks. Others had no discernible effect. For year-round corrosion protection, we see a strong case for using Biobor EB, Mercury Quickstor, Sea Foam, or ValvTect Ethanol.